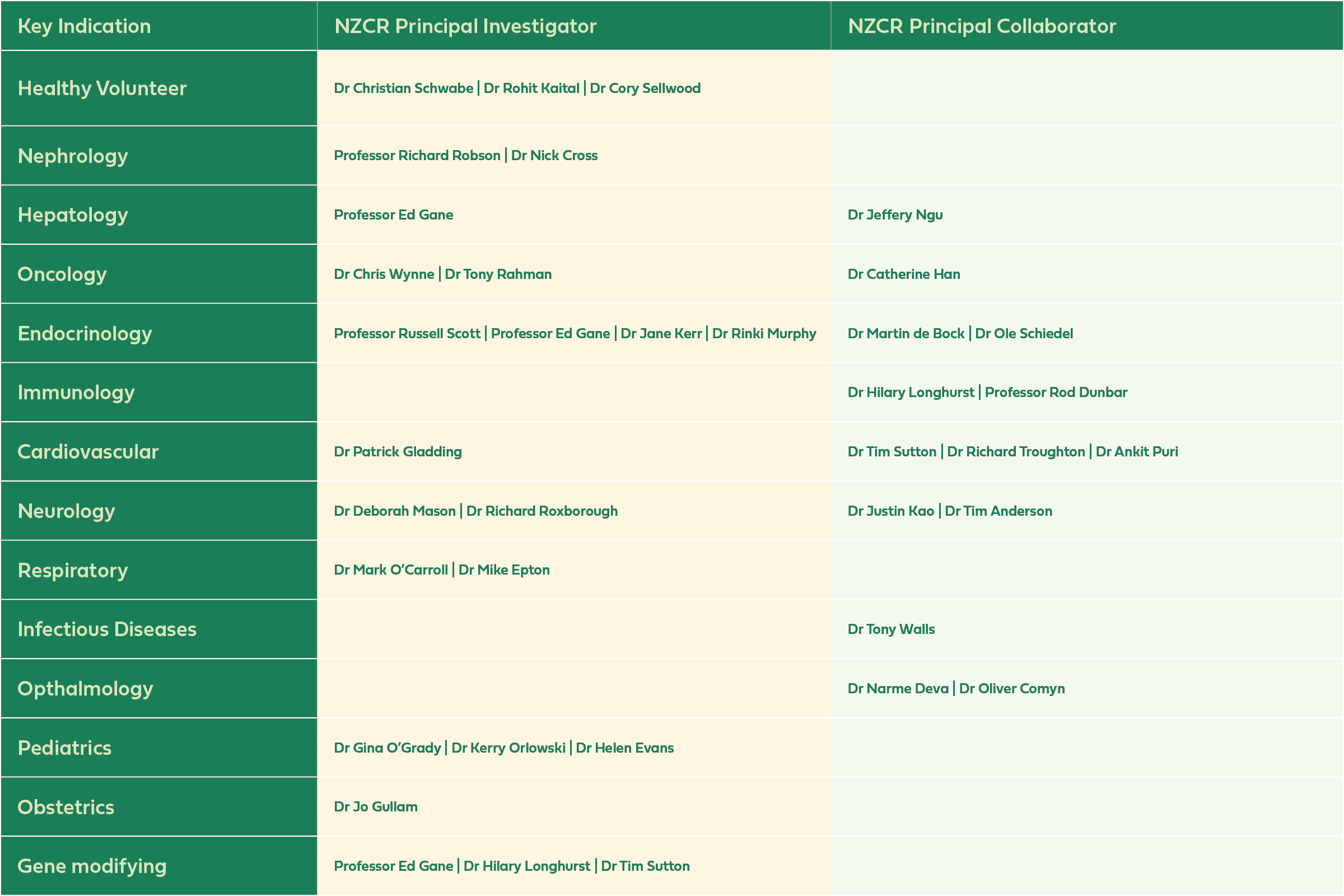
NZCR Research Highlights:
Our focus on quality and innovation has resulted in many exciting medical breakthroughs. This has included the development of therapies for many previously untreatable diseases, including hepatitis, cystic fibrosis, paroxysmal nocturnal haemoglobinuria, alpha-1-antitrypsin deficiency, and neuromuscular diseases.
Examples of our research highlights are:
-
- Major role in the development of a cure for chronic Hepatitis C
-
- Leading contributor to global efforts for a chronic Hepatitis B cure
-
- Conducted one of the largest global biosimilar studies involving 696 subjects
-
- Completed the initial dose-finding trial which led to Herceptin Sub cutaneous delivery
-
- First in world to develop umbrella protocols
-
- First in world in vivo Gene editing trials (SiRNA, mRNA delivery, CRISPR/CAS9)
Examples of NZCR publications:
Lawitz C; Landis CS; Flamm SL; Bonacini M; Ortiz-Lasanta G; Huang J; Zhang J; Kirby BJ; De-Oertel S; Hyland RH; Osinusi AO; Brainard DM; Robson RA; Maliakkal BJ; Gordon SC; Gane EJ. Sofosbuvir plus ribavirin and sofosbuvir plus ledipasvir in patients with genotype 1 or 3 hepatitis C virus and severe renal impairment: a multicentre, phase 2b, non-randomised, open label study. Lancet Gastroenteraol Hepatol 2020: June 9; 1-9.
Kirschbrown WP, Wynne C, Kågedal M, Wada R, Li H, Wang B, Nijem I, Badovinac Crnjevic T, Gasser H, Heeson S, Eng-Wong J, Garg Amit. Development of a Subcutaneous Fixed‐Dose Combination of Pertuzumab and Trastuzumab: Results From the Phase Ib Dose‐Finding Study Journal of Clin Pharm. 2019 May: 59(5):702-716.
Wright RS, Collins MG, Stoekenbroek RM, Robson R, Wijngaard PLJ, Landmesser U, Leiter LA, Kastelein JJP, Ray KK, Kallend D. Effects of Renal Impairment on the Pharmacokinetics, Efficacy, and Safety of Inclisiran: An Analysis of the ORION-7 and ORION-1 Studies. Mayo Clin Proc. 2020 Jan;95(1):77-89
Wynne C, Schwabe C, Vincent E, Schueler A, Ryding J, Ullmann M, Ghori V,Kanceva R, Stahl M. Immunogenicity and safety of a proposed pegfilgrastim biosimilar MSB11455 versus the reference pegfilgrastim Neulasta® in healthy subjects: A randomized, double-blind trial.Pharmacol Res Perspect. 2020 Apr;8(2):e00578.
Wynne C, Schwabe C, Sachdeva Batra S, Lopez-Lazaro L, Kankanwadi S. A comparative pharmacokinetic study of DRL_BZ, a candidate biosimilar of bevacizumab, with Avastin® (EU and US) in healthy male subjects. British Journal of Clinical Pharmacology. 2018 Oct:84(10):2352-2364.